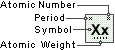Cesium
Symbol
Cs
Atomic Number
55
Atomic Weight
132.90543
Element Classification
Discovered By
Gustov Kirchoff, Robert Bunsen
Discovery Date
1860 (Germany)
Name Origin
Latin: coesius (sky blue), for the blue lines of its spectrum.
Density (g/cc)
1.873
Melting Point (K)
301.6
Boiling Point (K)
951.6
Appearance
Very soft, ductile, light gray metal
Atomic Radius (pm)
267
Atomic Volume (cc/mol)
70.0
Covalent Radius (pm)
235
Ionic Radius
167 (+1e)
Specific Heat
0.241
Fusion Heat (kJ/mol)
2.09
Evaporation Heat (kJ/mol)
68.3
Thermal Conductivity (@25°C W/m K)
35.9
Debye Temperature (K)
n/a
Pauling Negativity Number
0.79
First Ionizing Energy (kJ/mol)
375.5
Oxidation States
1
Electronic Configuration
[Xe] 6s1
Lattice Structure
Body-Centered Cubic(BCC)
Lattice Constant
6.050
Lattice C/A Ratio
n/a